Celiac Disease: Pathogenesis, Symptoms, and Lifelong Management
A comprehensive clinical overview of gluten-sensitive enteropathy and its systemic impact on the human body.
Understanding the Autoimmune Mechanism
Celiac disease — you may also see it called gluten-sensitive enteropathy or celiac sprue — is a severe and chronic autoimmune disorder. It's not a food allergy, which would trigger an immediate histamine response like the anaphylactic reaction to peanuts, and it's not a simple food intolerance that mostly causes localized digestive discomfort. Celiac disease is something more complex: when someone with this genetic predisposition eats gluten, their immune system misidentifies the protein complex as a dangerous pathogen. What follows is an aggressive inflammatory response — but it doesn't attack the gluten. It attacks the body's own small intestine.
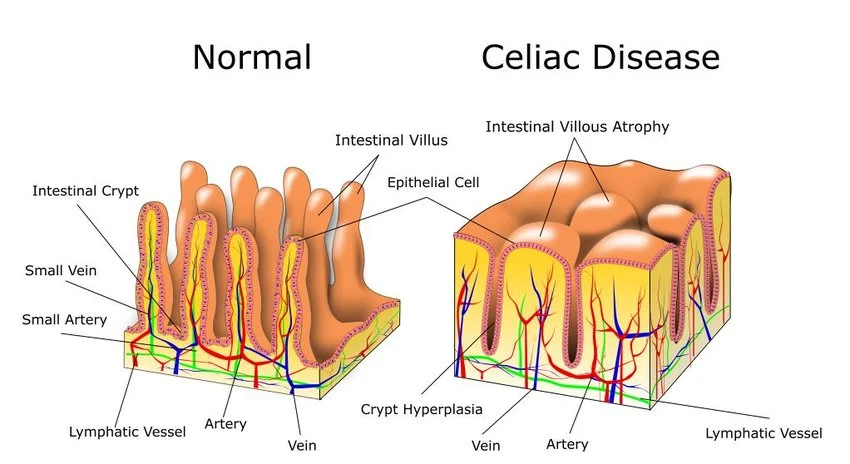
The main target of this autoimmune attack is the intestinal villi — the microscopic, hair-like projections that densely line the mucosal wall of the small bowel. Their job is to massively increase the intestine's surface area, enabling efficient absorption of macronutrients, vitamins, and minerals into the bloodstream. In someone with active Celiac disease, the sustained inflammatory response triggers a condition known as villous atrophy: the villi become severely inflamed, blunted, and eventually flatten completely. Once that happens, the intestine simply can't absorb nutrients properly, leading to profound systemic malnutrition no matter how much or how well you eat.
The Gluten Spectrum: Ubiquity and Cross-Contamination
Gluten is an umbrella term for a group of structural proteins — mainly gliadin and glutenin — found naturally in cereal grains, most notably wheat, barley, and rye. It's the ingredient that makes bread chewy and gives pasta and cakes their structure, which is exactly why it's so deeply embedded in modern food production.
But managing a strict gluten-free diet goes far beyond skipping bread and pasta. Because of its structural properties, the food industry routinely uses gluten as a binding agent, thickener, and flavor enhancer in processed foods. It shows up in soy sauces, malt vinegars, salad dressings, canned soups, and processed meats — and even outside the kitchen, in certain medications, vitamin supplements, and lip balms. Beyond hidden ingredients, people with Celiac disease also need to stay vigilant about cross-contamination. Food prepared on the same surfaces, fried in the same oil, or handled with the same utensils as gluten-containing items can carry enough trace gluten to set off a serious mucosal inflammatory response — even if the food itself appears to be gluten-free.
Clinical Presentation and Symptomatology
Celiac disease has a reputation in clinical circles as a chameleon. Its presentation is notoriously heterogeneous — symptoms can range from severe and debilitating to completely absent (so-called silent Celiac disease), even as significant intestinal damage accumulates in both cases.
Gastrointestinal Manifestations
The classic symptoms start in the gut, driven by the malabsorption of fats and carbohydrates. Patients frequently deal with chronic, persistent diarrhea marked by steatorrhea — stools that are bulky, pale, abnormally foul-smelling, and hard to flush because of their high undigested fat content. That's typically accompanied by severe bloating, intense colicky stomach pain, and flatulence. In some cases, the damaged gut motility slows down enough to cause severe constipation instead.
Extra-Intestinal Manifestations
Because the disease essentially destroys the body's absorption mechanisms, a wide array of symptoms emerge well outside the gut. Chronic fatigue is one of the most common complaints, primarily driven by iron-deficiency anemia — the damaged intestine can't absorb dietary iron properly. Skin is another target: dermatitis herpetiformis, an intensely itchy, blistering rash that appears on the elbows, knees, and buttocks, is a well-recognized dermatological manifestation. Neurologically, patients may experience peripheral neuropathy (tingling in the extremities), persistent migraines, and the cognitive impairment commonly called "brain fog." Left untreated, the condition can also lead to osteoporosis from calcium malabsorption, infertility, and serious developmental delays or stunted growth in children.
Diagnostic Protocols and the Importance of Tracking
Diagnosing Celiac disease typically starts with blood work. Physicians screen for specific autoantibodies — most commonly Tissue Transglutaminase IgA (tTG-IgA) and Endomysial antibodies (EMA). If those markers come back elevated, the diagnosis is confirmed with an upper gastrointestinal endoscopy, during which a gastroenterologist takes multiple biopsies from the duodenum (the first section of the small intestine) to directly assess the degree of villous atrophy under a microscope.
This is critical: don't go gluten-free before you've been tested. The diagnostic tests depend entirely on measuring your body's active immune response to gluten and observing the resulting mucosal damage. If you cut out gluten early, the gut starts to heal and antibody levels fall — which frequently leads to a false-negative diagnosis and leaves the real cause of your symptoms unidentified.
Once you've received a positive diagnosis, the only medically accepted treatment is strict, lifelong adherence to a gluten-free diet — no exceptions. In this phase, careful data collection becomes an important recovery tool. Apps designed to track digestive output, dietary intake, and daily habits let patients monitor their mucosal healing over time. By documenting shifts in stool consistency using the Bristol Stool Scale, logging the frequency of abdominal distress, and keeping precise dietary records, you can quickly spot accidental cross-contamination events and bring accurate, detailed reports to your gastroenterology follow-ups. For managing dietary triggers, the Low-FODMAP diet is another evidence-based protocol sometimes used alongside gluten avoidance.
Sources & Medical References: